Hello All!
The past few weeks we have had some great news in our laboratory we recently got word of manuscript acceptances for a paper by one of our former PhD students Jacob Brown, titled “Protein imbalance in the development of skeletal muscle wasting in tumor-bearing mice“, accepted to the Journal of Sarcopenia, Cachexia, and Muscle and one of our former Masters students Thomas Blackwell titled “A Transcriptomic Analysis of the Development of Skeletal Muscle Atrophy in Cancer-Cachexia in Tumor-Bearing Mice“ accepted to Physiological Genomics. Both papers came from a study we completed a couple years ago investigating how the muscle alters and changes before muscle wasting during cancer-induced muscle wasting.
Most people would agree cancer is still a significant health problem in the United States and the world, one side effect that accompanies many cancers is cancer-cachexia, essentially this is a loss of body weight, mostly muscle and fat mass that is caused by a combination of the cancer and the cancer treatment. This unintentional weight loss is problematic because many time cancer treatments are based on lean mass (e.g. muscle) and if someone has less muscle, doctors can’t use as aggressive cancer treatments, which can contribute to increased mortality. In fact, many studies have found that muscle mass size very closely relates to mortality and quality of life in these patients. However, what’s causing this loss of muscle mass is still unknown, the obvious answer may be to feed these patients more food, but we now know that just giving more food is not enough to reverse or prevent this cancer-induced cachexia. So our group wanted to know what may be happening after cancer has begun to develop, but before the muscle actually starts to shrink. Therefore, the purpose of this study was to look at how the progression of cancer relates to different measures of muscle health.
I’ll start with Jacob’s paper, where we were predominantly looking at what we call “protein turnover”. Our muscle size is a combination of making proteins (protein synthesis) and breaking down proteins (protein degradation). When protein synthesis is larger than protein degradation (such as when someone starts a resistance exercise training program) we get larger muscles, however when protein degradation is larger than protein synthesis, we get smaller muscles (this is a common occurrence with very long periods of bed rest). What we wanted to know for this study was how these two processes change during cancer progression to contribute to muscle loss. To do this we looked at muscle samples at 0, 1, 2, 3, and 4 wks of cancer progression. We found that by 3wks-4wks of cancer progression muscle cross-sectional area (a measure of muscle size) was significantly smaller, but protein synthesis did not decrease until 4wks of cancer progression. Additionally, we also found increases in measures of protein degradation that corresponded with the muscle wasting, suggesting both a decrease in making proteins and an increase in breaking down proteins are contributing to muscle loss during cancer cachexia progression. Importantly, we did not find any alterations in these processes BEFORE muscle actually began to shrink. We have previously found that muscle mitochondria become unhealthy very early during cancer progression (see previous blog post), therefore these two studies together suggest that cancer causes muscle mitochondria to become unhealthy very early which may then contribute to reduced protein synthesis and increased protein breakdown, which then contributes to smaller muscles and overall reduced quality of life.
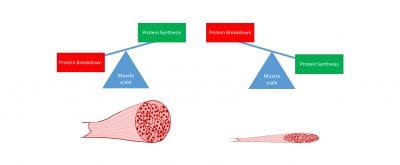
How we gain and lose muscle, if we have more protein making (synthesis) our muscles will be larger, however if we have more protein breakdown our muscles will be smaller.
For Thomas’s paper we used samples from the same study to look a gene alterations using gene sequencing. We all have genes, these are given to use by our parents, and our genes provide the blue print for our body to make proteins. Our gene “blue print” is called RNA, if our body needs more or less of a certain type of protein, we will start mechanisms to increase or decrease our “blue prints” (RNA). Therefore, we can measure the content of different RNAs and determine how the body is trying to regulate itself. Now, we have thousands of genes, which means thousands of potential RNA “blue prints” to measure, making analyzing each gene individually essentially impossible. However, a new technique was recently developed called “transcriptomics”, where essentially, we take a sample and look at all of the RNAs in a sample and if they are increased or decreased relative to control samples (some may refer to this as a “shot gun” approach). This gives us a HUGE data set and the ability to look at different processes that may have never been investigated in cancer research. For this study we did this type of analysis during 0, 1, 2, 3, and 4 wks of cancer progression. We found that most of the differences in RNA content occurred after 4wks of cancer progression relative to control, specifically, we saw large alterations in genes related to mitochondria dysfunction and protein synthesis/degradation. These findings align with our other works, more so, these suggest that alterations of the RNA become problematic at 4wks. This is important, because RNA is the precursor to proteins and therefore the precursor to almost every cellular process that occurs in our bodies. If RNA alterations occurred very early in cancer progression (such as 1wks), this would suggest that stopping/reversing cancer-induced wasting is very difficult if not impossible. Because some of these changes did not occur until 4wks (recall this also about the time the muscle is smaller) this suggests that it may be possible to stop cancer-induced wasting before the muscle wastes. Which could be a very important finding and influence how we are develop cancer treatments.
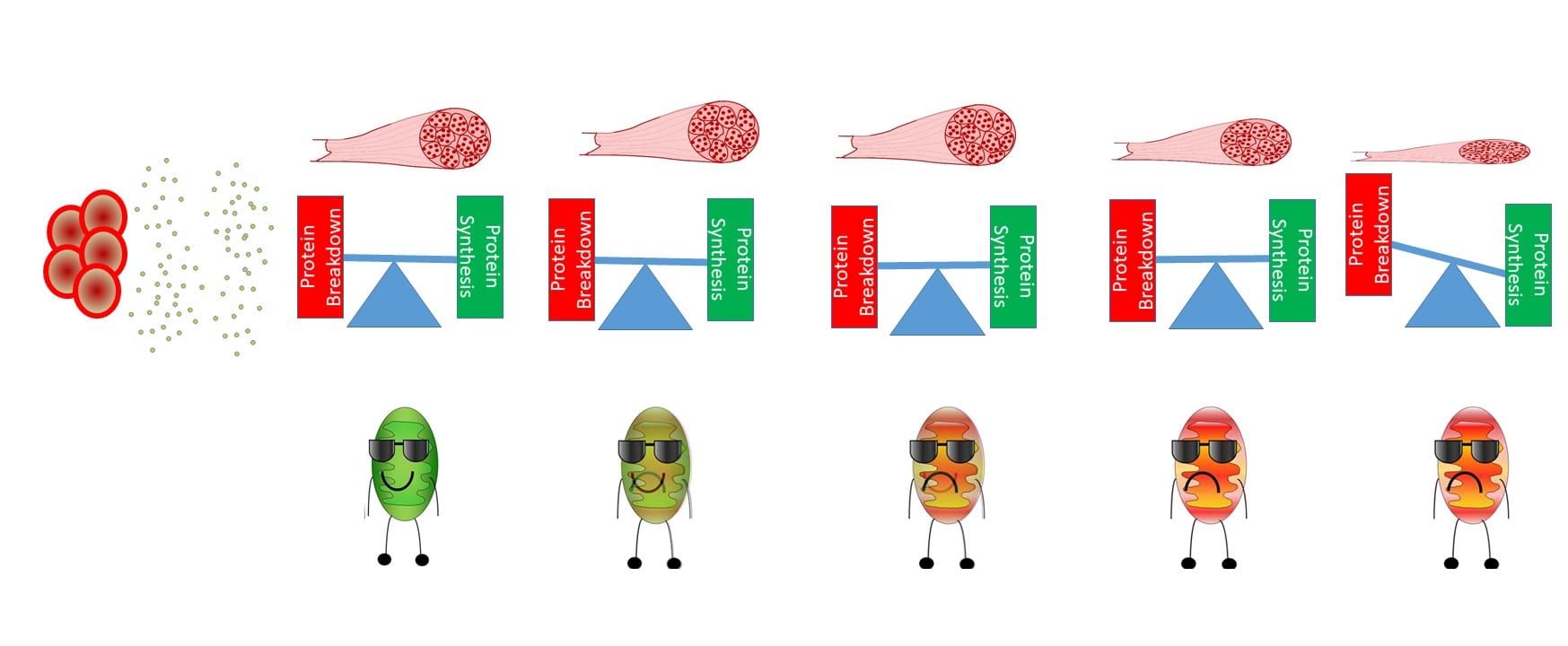
A quick pictorial description of some of the things we have found! With cancer progression mitochondria become less healthy, but this doesn’t manifest in muscle wasting until after cancer has progressed for awhile.
Now with any study there are a few limitations I want to quickly address. Both of these studies were conducted in male mice, therefore more work is needed to understand if these same processes occur in female mice and humans in general. Additionally, this is one type of cancer, as many cancer scientists or oncologists will attest, different cancers can behave very differently, so more work will be needed to see if these findings are similar in other cancer models.
We just got back from Central States ACSM and had a great meeting. Look forward to it for our next post! Until then keep on #sciencing!
Cheers!
Megan